Mandala #122 - Mandala Madness - Art, Abstract, Soul, Color, Life, Body, Peace, Generative, Love, Dream, Buddha | OpenSea
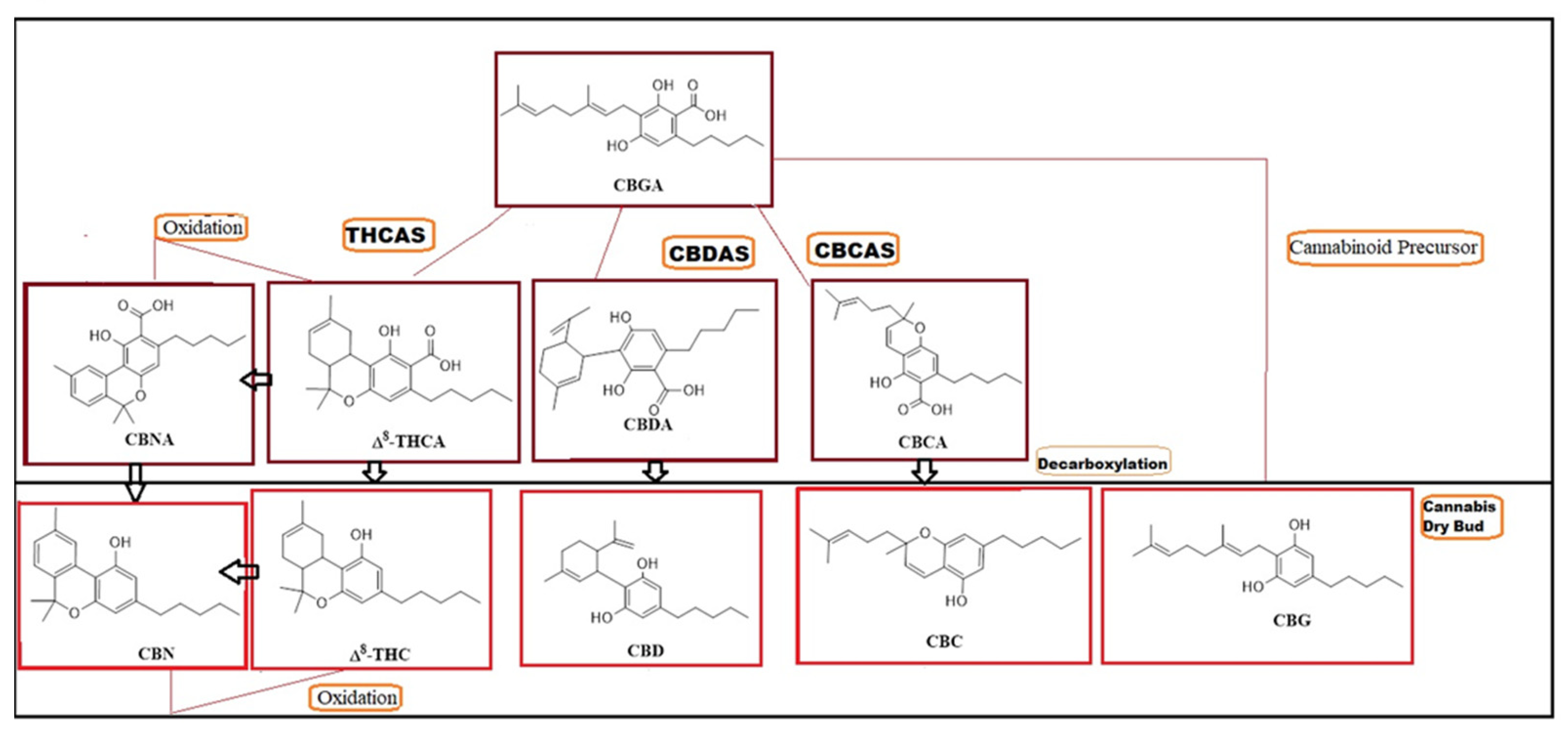
Molecules | Free Full-Text | Post-Harvest Operations to Generate High-Quality Medicinal Cannabis Products: A Systemic Review

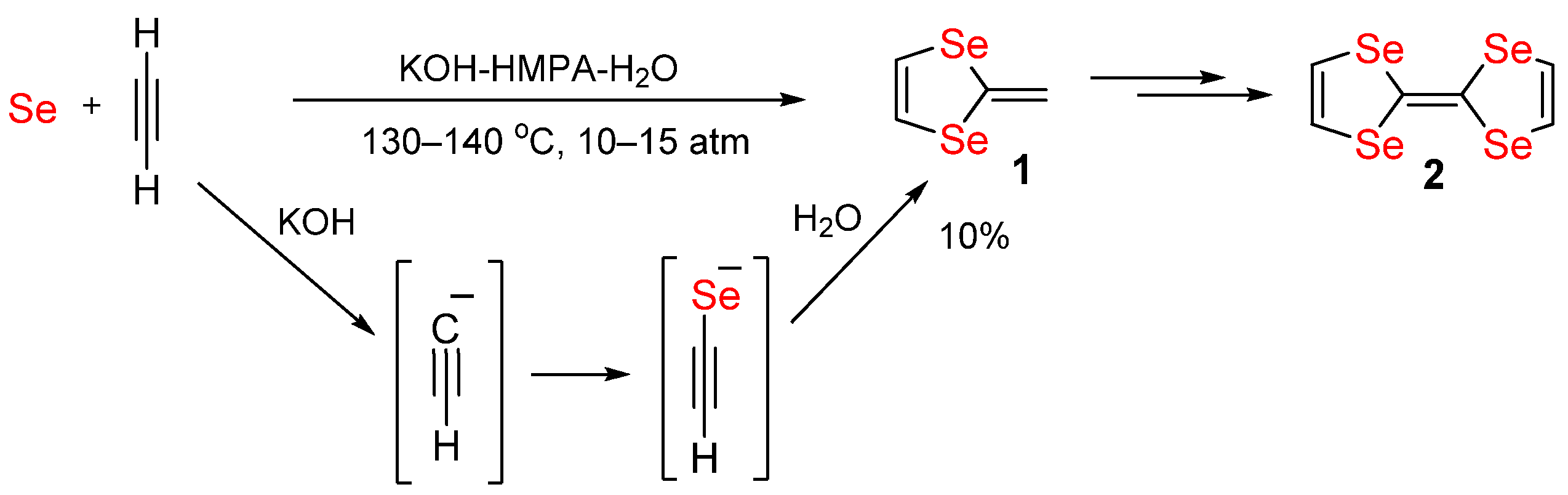
Sharp difference in the rate of formation and stability of the Diels–Alder reaction adducts with 2,3‐dicyano‐1,4‐benzoquinone and N‐phenylimide‐1,4‐benzoquinone‐2,3‐dicarboxylic acid - Kiselev - 2021 - International Journal of Chemical Kinetics - Wiley ...

Porphyrindiene-Based Tandem Diels–Alder Reaction for Preparing Low-Symmetry π-Extended Porphyrins with Push–Pull Skeletons | The Journal of Organic Chemistry